Calculating formal charge co11/28/2023 Since sulfur has six valence electrons, we conclude that two electrons are not involved in the bonding, i.e., that there is a lone pair. In the sulfite ion, SO 3 2– for example, the oxidation number of sulfur is +4, suggesting that only four sulfur electrons are involved in the bonding. The formula for calculating the formal charge on an atom is simple. Oxidation numbers can sometimes also be useful in writing Lewis structures, particularly for oxyanions. 
I/We hereby authorise Shubham Housing Development Finance Co. For calculating the formal charge, you have to use the following formula Formal. Shubham is a leader in providing formal housing to those with informal incomes. 
This type of reaction can be recognized because it involves a change in oxidation number of at least one element. How to Calculate the Formal Charges for CO (Carbon Monoxide) Wayne Breslyn. Oxidation numbers are mainly used by chemists to identify and handle a type of chemical reaction called a redox reaction, or an oxidation-reduction reaction. I) Cr(IV) j) Cu(II) k) Fe(IV) Answer Answer aĬoordination complexes most often have 18 electrons on the metal atom.\,\] 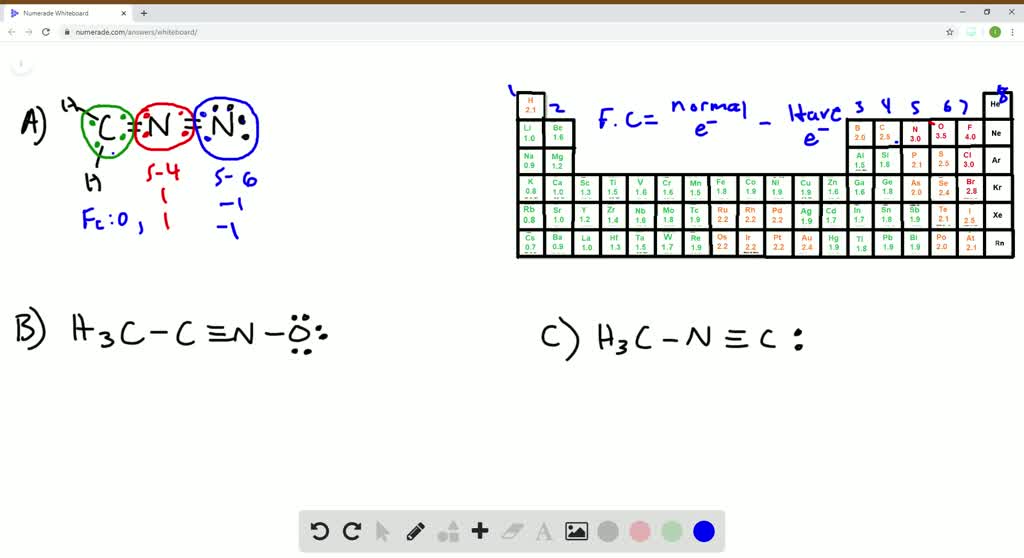
How many valence electrons are found on the following metal ions? A) CO2 The Lewis structure for CO2 is There are two other resonance. In cases like this one, the oxidation state is the same thing as the charge on an ion. 2: Carbon tetrachloride has zero dipole moment since. In main group, covalent compounds, we always adjust the electron count to reflect an overall charge on the structure. So the formal charge of carbon monoxide (CO) is zero. A further wrinkle occurs if there is an overall charge on the complex. Therefore the net formal charge on the carbon monoxide - 1 + 1 0. 3: Counting electrons on the anionic ligands and the metal ion. Oxidation state refers to how many electrons an atom has lost (or gained). The formula used to calculate formal charge. The total electron count on the metal in the complex is sixteen. In contrast to the formal charge, in which the electrons in a bond are assumed to be shared equally. This number is formally called the oxidation state. The formal charge FC is then calculated by subtracting NBE and B from VE. counting the number of bonds (B) to the atom, or alternatively, counting the number of bonding electrons and dividing this by 2. 
charge any family, the reimbursement amount and the State assigned co-pay. Complete answer: - In the question it is asked what the formal charge of CO is. \): Some bases commonly used in synthetic chemistry.įor each complex in the previous problem, what is the valence electron count at the metal in the complex? Answer aĬharge on metal is often represented as a Roman number in parentheses. Each lone pair counts as 2, and each unpaired electron counts as 1. Full, formal scholarship - can be used for care provided by a Maryland registered. Hint: The formal charge of the atom in a polyatomic molecule is going to depend on the number of electrons gained from other atoms or donated to other atoms.The formal charge of the atom in the molecule will be positive or negative or neutral.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
